Testing’s Role in Preventing HIV Infection
About 1.2 million people in the United States are HIV-positive. One in five of those don’t know they’re infected, a major contributing factor to the 50,000 or so new HIV infections a year. Testing is the first step to engage people with HIV in care, stop individual disease progression, and stem the spread of HIV. In accordance with CDC recommendations, NCCC consultants advise that providers test all patients aged 13 to 64, regardless of risk profile, at least once in their lifetime, preferably annually as part of routine health screening.
See our PrEP Resources page for information on the use of pre-exposure prophylaxis (PrEP) as a prevention tool.
Frequently Asked Questions about HIV Screening and Testing
Updated September 2025
Information and resources to aid in implementing routine testing in your practice.
Provisional Guidance Notice:
Updated HIV screening and testing guidance is being shared to reflect recent changes to clinical practice recommendations. The HRSA review and clearance process is currently experiencing delays; final content will be posted once the review and approval process is complete.
Disclaimer:
Testing recommendations and technology change over time, and the introduction of HIV pre-exposure prophylaxis (PrEP)—particularly long-acting injectable PrEP—has brought new challenges to HIV test interpretation. Please refer to the CDC website and other resources for updated information, or call 1-844-ASK-NCCC (844-275-6222) for expert guidance on interpreting test results.
What are the current recommendations for HIV screening/testing?
One-time HIV screening for all persons aged 15-65 is recommended by the US Preventive Services Task Force (USPSTF), as part of regular medical care; the Task Force also recommends screening for younger adolescents and older adults with potential HIV exposures. The CDC recommends one-time routine screening for all persons aged 13-64.
- USPSTF states it is “reasonable to consider” repeat screening with potential ongoing HIV exposures, although they note there is insufficient evidence to make specific recommendations on the frequency or interval of such screening.
- CDC recommends at least annual screening for people with potential ongoing HIV exposures.
- USPSTF and CDC both recommend screening all pregnant persons, including those who present in labor or at delivery with unknown HIV status. CDC also recommends repeat screening during the third trimester for pregnant women with risk factors for HIV acquisition and in women living or receiving care in high-incidence settings. Repeat screening is also recommended for all pregnant persons who previously tested negative during each subsequent pregnancy.
HIV screening tests are administered on an “Opt-out” vs. “Opt-in” basis. “Opt-out” consent refers to general informed consent for medical care, notifying the individual that an HIV test will be performed unless they decline, and should be considered adequate informed consent for HIV testing.
- This routine screening approach does not require specific pre-test counseling or separate written consent* as had been the practice for “Opt-in” testing.* State-specific variations in HIV testing laws may exist: https://www.usa.gov/state-health.
Are there additional considerations for persons with potentially frequent HIV exposures?
HIV acquisition occurs more frequently in areas/populations with high HIV prevalence, and specific scenarios are also associated with a higher likelihood of HIV exposure. More frequent testing (e.g., annually or more often) should be offered, and may be guided by estimated exposure frequency, to:
- People who have recently been diagnosed with a bacterial sexually transmitted infection (STI);
- People who inject drugs and share injection-related equipment;
- People having condomless sex and/or who are not on HIV pre-exposure prophylaxis (PrEP) and whose partners are living with untreated HIV, or whose partners are receiving HIV treatment but experiencing viremia;
- People having condomless sex and/or who are not on PrEP who have sex partners of unknown HIV status; and
- People who are being evaluated for HIV pre- or post-exposure prophylaxis, as well as those receiving treatment for viral hepatitis or tuberculosis.
What types of HIV tests are available?
There are 3 basic types of HIV tests: HIV antigen/antibody (HIV Ag/Ab) combination tests; HIV antibody (IgM and/or IgG) tests; and HIV nucleic acid tests (NATs). Both HIV antigen/antibody combination tests and HIV antibody tests have options for laboratory-based (“instrumented”) testing and “point-of-care” based testing. Other types of HV diagnostic tests that are available include HIV-1/2 differentiation assays and a home-based HIV antibody test.
What are the differences between various types of HIV tests?
One key difference between the 3 types of tests is the “window period” for HIV infection detection, with HIV nucleic acid tests having the shortest detection interval between potential HIV acquisition and a reliable test result (see additional details in the “window period” section, below). Additional differences are described below.
- Laboratory-based (instrumented) HIV Ag/Ab tests: detect both HIV-1 p24 antigen and IgM/IgG antibodies to HIV-1 and HIV-2. Because this approach can detect HIV earlier than any of the other types of Ag/Ab or Ab-only tests, instrumented Ag/Ab assays are generally the preferred screening tests for HIV. Multiple such assays are FDA-approved and have slight differences in test characteristics and performance. Turnaround time varies but almost all can be completed in 1 hour or less.
- HIV Ag/Ab point of care tests: also detect both HIV-1 p24 antigen and IgM/IgG antibodies to HIV-1 and HIV-2. These assays use lateral flow immunochromatographic technology (similar to what is used for a urine pregnancy test, for example), and are not as sensitive at identifying acute or recent HIV infection compared to laboratory-based HIV Ag/Ab assays. Results are available in 20 minutes, so these may be useful when an immediate result is needed (e.g. for pregnant people presenting in labor, HIV post-exposure evaluations, and/or when rapid antiretroviral therapy/PrEP is being considered).
- HIV Ab laboratory-based tests: most currently available laboratory-based Ab assays detect both HIV IgM and HIV IgG antibodies; IgM antibodies can typically be detected ~ 3-4 weeks after HIV acquisition. However, because of the longer window period of these assays compared to the combined Ag/Ab assays, these are no longer considered the initial screening test of choice.
- HIV Ab point of care tests: use immunochromatographic flow technology to detect antibodies to HIV-1 +/- HIV-2 (depending on the assay). Results are typically available within 30-40 minutes, so these may also be useful when an immediate result is needed (see above). However, they are less sensitive than laboratory-based HIV Ag/Ab tests and require additional testing to confirm HIV when results are positive/reactive.
- HIV nucleic acid tests (NAT): detect HIV-1 and/or HIV-2 RNA or DNA; may be qualitative (result reported as positive/detected or negative/not detected) or quantitative (result reported as a specific number). Currently, there are 3 HIV NAT assays that are FDA-approved for HIV diagnosis.
- HIV-1/2 Ab differentiation assays: detect specific antibodies to both HIV-1 and HIV-2. Based on which antibodies are detected, enables differentiation between the two types of HIV infection.
- HIV in-home tests: facilitate self-testing outside of traditional clinical settings and do not require specific professional training or whole blood for sample collection. There are currently 2 “in-home” tests available—one (saliva-based) can be performed entirely at home, with results in 20 minutes; the other uses a finger-stick blood sample collected at home that must be mailed in to the manufacturer for processing. Both have lower sensitivity for diagnosing HIV than other available assays, particularly for acute or recently acquired HIV.
How is a diagnosis of HIV established?
An HIV diagnosis can be established when a positive HIV screening test is followed by a positive supplemental test when following the CDC’s recommended algorithm (see figures below). One key exception to this is acute HIV infection, which can generally be established with a positive HIV NAT in someone with a negative HIV screening assay in the setting of a recent HIV exposure. For people with recent or current PrEP use, ambiguous or fluctuating HIV test results have been observed and therefore expert guidance (844-ASK-NCCC/844-275-6222) may be helpful in assessing whether someone may have acquired HIV in the context of PrEP use.
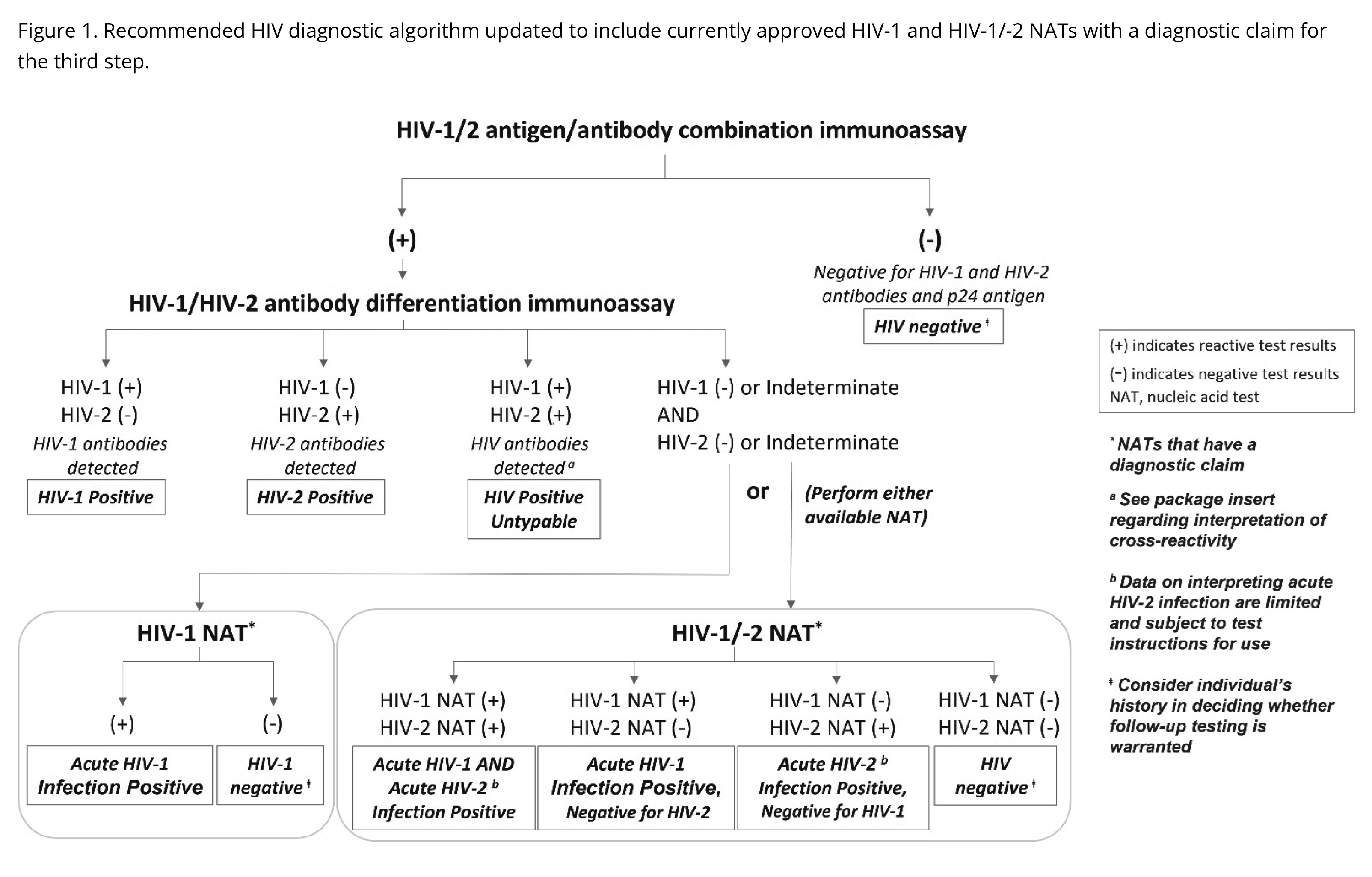
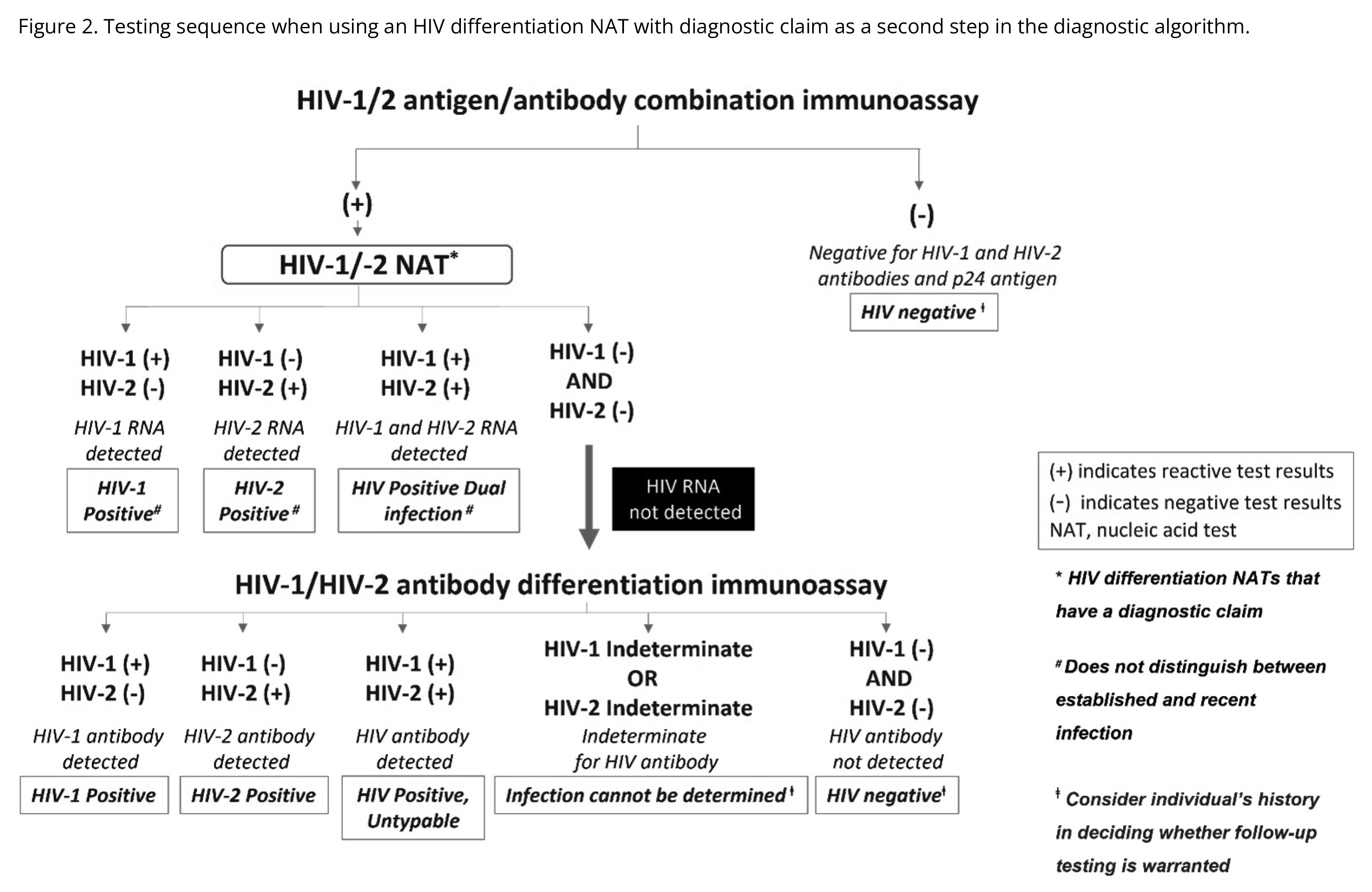
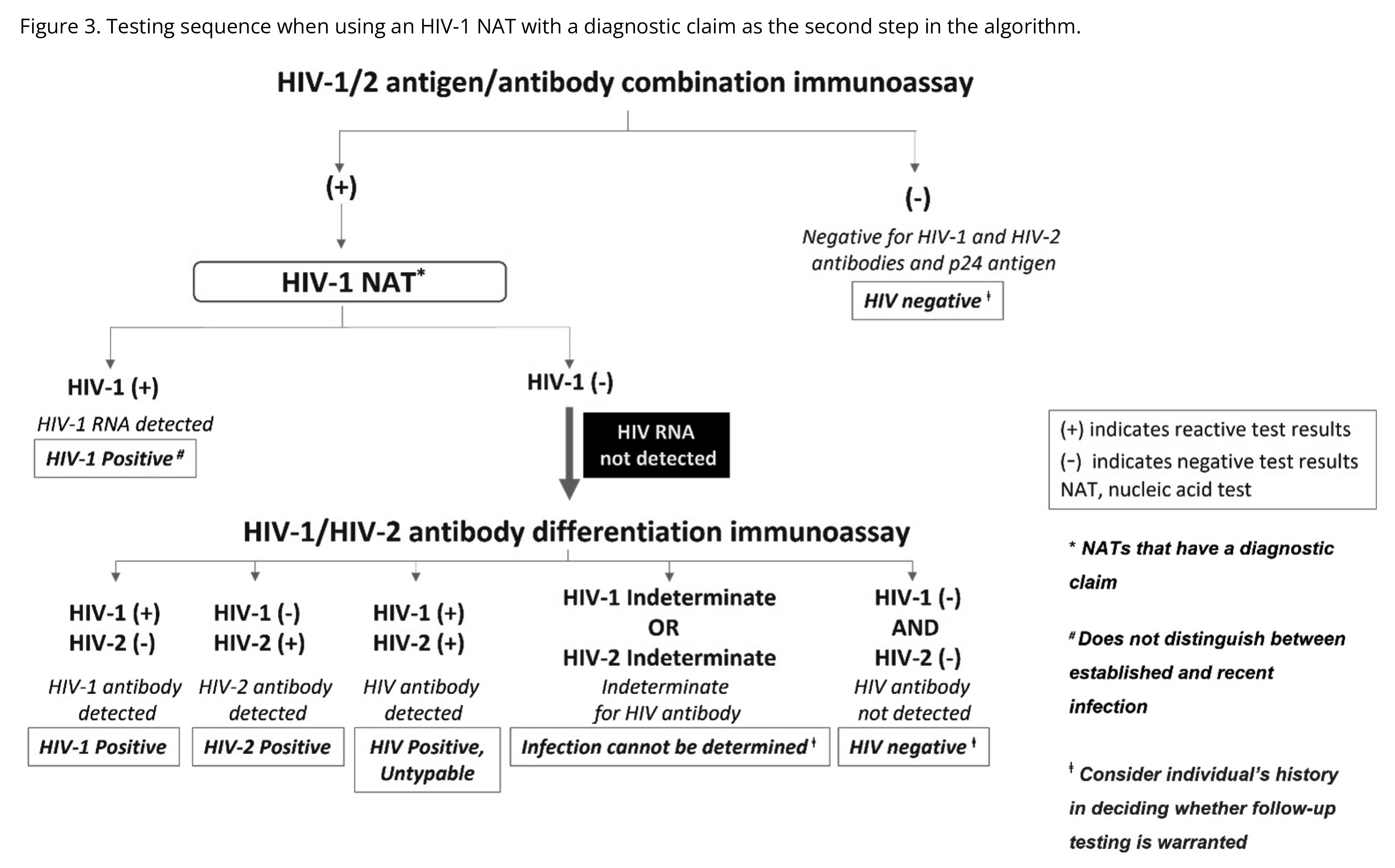
Figures 1-3 from https://stacks.cdc.gov/view/cdc/129018
What are the causes of false negative/false positive HIV screening tests?
False negative results are most often seen during the window period of infection (see below); they can also occur due to laboratory error, recent and/or ongoing exposure to antiretroviral therapy (ART) (e.g. HIV pre- or post-exposure prophylaxis or infant postnatal antiretroviral prophylaxis). They can also occur in rare clinical scenarios involving the inability to generate an adequate HIV antibody response (such as with hypogammaglobulinemia, or receipt of certain immunosuppressive medications).
False positive results may occur because of laboratory error, or the presence of cross-reactive/ interfering antibodies which can be produced in certain conditions (e.g., pregnancy, autoimmune diseases, certain viral infections) or after receipt of certain vaccines such as the influenza vaccine.
What is the “window period” and how does it impact HIV screening/testing results?
The window period is the time between HIV acquisition/infection and when a test result becomes reliably positive. The window period is shortest for HIV nucleic acid tests (on average 7-14 days), followed by p24 antigen (10-21 days), and then longest for HIV antibodies (2-8 weeks). During the window period, a person is infected with HIV but their laboratory results may not indicate infection.
The window period is significant because:
- The HIV viral load level is usually high during the first several weeks and months after acquisition, if suppressive ART is not initiated early. As a result, there is an increased risk for transmitting HIV to others.
- People who receive a positive HIV diagnosis during the window period should be quickly referred to follow-up care and treatment, which can improve health outcomes.
Resources
U.S. Centers for Disease Control and Prevention (CDC)
Retrieved from: https://www.cdc.gov/hivnexus/hcp/diagnosis-testing/index.html#cdc_hcp_diagnosis_recommended-recommended-tests
US Preventive Services Task Force (USPSTF), Screening for HIV
Retrieved from: https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/human-immunodeficiency-virus-hiv-infection-screening
National HIV Curriculum, HIV Diagnostic Testing
Retrieved from: https://www.hiv.uw.edu/go/screening-diagnosis/diagnostic-testing/core-concept/all
Linkage to Care
Information on linking your patients to appropriate testing resources and care.
- AIDS Education and Training National Coordinating Resource Center Resource Library
 Open Link | National AETC Support Center (NASC)
Open Link | National AETC Support Center (NASC)
- AIDS Education and Training Regional Centers
 Open Link | National AETC Support Center (NASC)
Open Link | National AETC Support Center (NASC)
- HIV Medicine Association Provider Directory
 Open Link | HIV Medicine Association (HIVMA)
Open Link | HIV Medicine Association (HIVMA)
- Find HIV Services Locator
 Open Link | U.S. Centers for Disease Control and Prevention (CDC)
Open Link | U.S. Centers for Disease Control and Prevention (CDC)
- CDC: Find a Testing Site
 Open Link | U.S. Centers for Disease Control and Prevention (CDC)
Open Link | U.S. Centers for Disease Control and Prevention (CDC)
 University of California, San Francisco |
University of California, San Francisco |
